Polylaminine: The Brazilian Molecule That May Regenerate the Spinal Cord and Restore Movement to Paraplegics
Date: March 6, 2026
Reading time: 28 minutes
Emoji: 🧬
Imagine a world where a spinal cord injury — currently considered irreversible — could be repaired by a synthetic molecule that rebuilds the destroyed neural pathways. This is not science fiction. It's called Polylaminine, and it was created in Brazil. Researcher Alexandre Fogaça Cristante, from the Institute of Orthopedics and Traumatology at the Hospital das Clínicas of the University of São Paulo (HC-USP), has developed a biopolymer that has already demonstrated the ability to regenerate nerve fibers in animal models — and the first human trials are closer than ever. This article dives deep into the science, the journey, and the revolutionary potential of Polylaminine to transform the lives of millions of people worldwide.
What Is Polylaminine?
Polylaminine is a synthetic biopolymer derived from laminin, a glycoprotein that naturally exists in the basement membrane of human cells. Laminin plays a fundamental role in cell adhesion, tissue differentiation, and crucially, in the growth and guidance of axons — the extensions of neurons responsible for transmitting electrical signals throughout the nervous system.
Natural Laminin: The Inspiration
Laminin is one of the most studied proteins in neuroscience. It composes the so-called extracellular matrix — the molecular "scaffold" that supports and guides cell growth. In the nervous system, laminin is especially important during embryonic development, when neurons need molecular "rails" to grow in the right direction and form the synaptic connections that constitute a functional nervous system.

The problem is that in the adult central nervous system (brain and spinal cord), the natural capacity for regeneration is extremely limited. When a spinal cord injury occurs — from trauma, disease, or vascular accident — axons are destroyed, and the body forms a glial scar at the injury site. This scar, composed of cells called reactive astrocytes, functions as a physical and chemical barrier that prevents any attempt at neural regeneration.
The Innovation: Turning Laminin Into a Polymer
What Dr. Cristante and his team achieved was transforming laminin — normally a fragile and difficult-to-handle protein — into a stable, implantable polymer. Polylaminine is designed to:
- Serve as a bridge: Fill the gap created by the injury, providing a physical substrate for axons to grow through the damaged area
- Guide growth: Polylaminine's binding domains attract axons and direct them in the correct orientation — from top to bottom in the spinal cord
- Neutralize inhibitors: The molecule interacts with inhibitory signals from the glial scar, reducing their ability to block axonal growth
- Be biocompatible: Because it's derived from a human protein, Polylaminine doesn't trigger significant immune responses
The Creator: Alexandre Fogaça Cristante
Dr. Alexandre Fogaça Cristante is an orthopedic surgeon, associate professor at the University of São Paulo School of Medicine, and researcher at the Medical Investigation Laboratory in Orthopedics and Traumatology (LIM-41) at HC-USP. His journey in spinal cord injury research began in the early 2000s, when he was struck by the complete absence of effective treatments for patients with paraplegia and tetraplegia.
The Research Journey
| Period | Milestone |
|---|---|
| 2003-2008 | Initial research on the role of laminin in neural regeneration |
| 2008-2012 | Development of the laminin polymerization process |
| 2012-2015 | First in vitro tests (laboratory cell cultures) with promising results |
| 2015-2018 | Tests in murine models (rats) with complete spinal cord injury |
| 2018-2021 | Tests in larger animal models (pigs and non-human primates) |
| 2021-2024 | Formulation refinement and pre-clinical toxicity studies |
| 2024-2026 | Regulatory preparation for human clinical trials |

Over more than two decades of research, Cristante not only developed the molecule but built a profound understanding of the mechanisms that prevent neural regeneration — and how to circumvent them. "The spinal cord doesn't regenerate not because neurons don't want to grow," he explained in an interview with Folha de S. Paulo in 2025, "but because the environment around the injury is actively hostile to growth. Polylaminine changes that environment."
The Science Behind It: How Polylaminine Works
To understand how Polylaminine acts, it's necessary to understand what happens when the spinal cord suffers a traumatic injury — and why the body is unable to repair it naturally.
What Happens After a Spinal Cord Injury
When the spinal cord is damaged — whether from a car accident, a fall, a dive into shallow water, or any other trauma — a cascade of devastating biological events occurs:
- Acute phase (0-72h): The mechanical injury directly destroys neurons and axons at the point of impact. Hemorrhage, edema, and inflammation spread, causing additional damage to surrounding tissues (secondary injury)
- Subacute phase (3-14 days): The immune system attacks cellular debris. Macrophages and microglia infiltrate the area, releasing inflammatory substances that, paradoxically, can cause more damage than the original injury
- Chronic phase (weeks to months): The glial scar forms — a dense barrier composed of reactive astrocytes and inhibitory molecules such as chondroitin sulfate proteoglycans (CSPGs), Nogo-A, and MAG. This scar is virtually impenetrable to growing axons
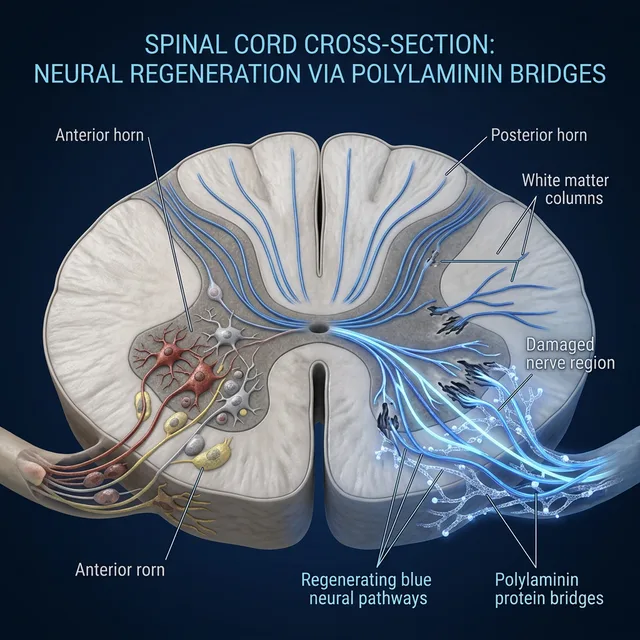
The Mechanism of Action of Polylaminine
Polylaminine acts on multiple simultaneous fronts:
1. Molecular Bridge
The biopolymer is injected directly at the injury site, where it forms a three-dimensional matrix that fills the gap between the intact segments of the spinal cord. This matrix functions as a bridge, providing physical support for axons to grow through the damaged tissue.
2. Growth Signaling
The integrin-binding domains present in Polylaminine activate receptors on neuron surfaces, triggering intracellular signaling cascades that promote:
- Axonal extension: Growth cones at the tips of axons are stimulated to advance
- Myelin production: Oligodendrocytes (cells that produce the myelin sheath) are recruited to coat the new axons
- Synapse formation: Once axons cross the injury, they establish new synaptic connections with neurons on the other side
3. Glial Scar Modulation
Perhaps the most innovative aspect of Polylaminine is its ability to modify the behavior of reactive astrocytes. Instead of simply trying to cross the scar, the molecule partially "reprograms" astrocytes, causing them to produce fewer inhibitory substances and more neurotrophic factors — substances that nourish and protect neurons.
4. Neuroprotection
Studies in animal models demonstrated that Polylaminine also exerts a neuroprotective effect: when administered in the first hours after injury, it reduces the extent of secondary damage, preserving neurons and axons that would otherwise be lost to inflammation.
Test Results: From Bench to Organism
The results obtained with Polylaminine over more than a decade of testing are, in the assessment of international experts, exceptional for a field where any progress is celebrated as revolutionary.
In Vitro Tests
In cell cultures, Polylaminine demonstrated:
- 300% increase in average axon length compared to untreated controls
- Directional growth: Axons grew preferentially along Polylaminine fibers, demonstrating guidance capability
- Complete biocompatibility: Zero cell toxicity at therapeutic concentrations
- Resilience: The polymer maintains its bioactive properties for weeks under physiological conditions
Tests in Murine Models (Rats)
Tests in rats with complete spinal cord injury (total cord transection) produced results that generated standing ovations at scientific conferences:
| Group | Motor Outcome (12 weeks) | Axonal Regeneration |
|---|---|---|
| Control (no treatment) | 0-1 on BBB scale (no movement) | 0% |
| Acute Polylaminine (injection < 24h) | 8-11 on BBB scale (wide joint movement) | 40-55% of axons regenerated |
| Chronic Polylaminine (injection > 4 weeks) | 4-6 on BBB scale (moderate joint movement) | 15-25% of axons regenerated |
The BBB scale (Basso, Beattie, Bresnahan) is the gold standard for evaluating motor recovery in spinal cord injury models. A score of 0 means complete paralysis; 21 means normal locomotion. Achieving scores of 8-11 in animals with complete spinal cord transection — where absolutely no spontaneous recovery is expected — is a result many consider the strongest credentials ever presented by any regenerative therapy in development.
Tests in Large Animal Models
The most recent results, presented at international conferences between 2023 and 2025, involved porcine and non-human primate models. Although complete data is still being published, preliminary results indicate:
- Partial functional recovery in primates with incomplete spinal cord injury
- No serious adverse effects in all treated animals
- Persistence of results for months after treatment, suggesting that regeneration is not transitory
International Adoption: The World Is Watching
Research with Polylaminine is not confined to Brazil. Dr. Cristante's work has attracted the attention of research centers and regulatory institutions across multiple continents.
International Collaborations

| Country/Institution | Type of Collaboration | Status in 2026 |
|---|---|---|
| USA — Johns Hopkins University | Parallel pre-clinical tests | Advanced stage |
| Germany — Max Planck Institute | Molecular mechanism analysis | Joint publications |
| Japan — Kyoto University iPS Cell Research | Combination with iPSC stem cells | Early studies |
| Israel — Weizmann Institute | Computational models of regeneration | Simulations underway |
| Canada — Toronto Western Hospital | Surgical protocol for implantation | Under development |
| UK — Cambridge Neuroscience | Advanced toxicology trials | Completed |
Regulation and the Path to Human Trials
The regulatory pathway for innovative biotechnological therapies is long and rigorous — for good reasons. Patient safety is always the top priority. In the case of Polylaminine, the process follows several stages:
- Complete pre-clinical studies (toxicology, pharmacokinetics, efficacy in multiple species) — ✅ Completed
- Submission to ANVISA (Brazilian National Health Surveillance Agency) — 🔄 In progress
- Submission to FDA (Food and Drug Administration, USA) — 🔄 Preparation
- Phase I Clinical Trial (safety in humans, 10-20 patients) — Expected 2027
- Phase II Clinical Trial (preliminary efficacy, 50-100 patients) — Expected 2028-2029
- Phase III Clinical Trial (large-scale efficacy, 200-500 patients) — Expected 2030-2032
ANVISA granted Polylaminine Advanced Therapy status, a classification that accelerates the regulatory process without compromising safety standards. The American FDA has also shown interest in granting Breakthrough Therapy designation, which would allow more frequent dialogue between researchers and the regulatory agency.
The Global Context: Spinal Cord Injuries in Numbers
To comprehend the potential impact of Polylaminine, it's essential to understand the global dimension of the problem it seeks to solve.
Numbers That Impress
- 250,000 to 500,000 people suffer new spinal cord injuries each year worldwide, according to the WHO
- More than 27 million people live with spinal cord injury consequences globally
- Treatment cost: $1-4.7 million per patient over a lifetime (in the USA)
- Average age: 30-40 years — affecting people at the peak of their productive lives
- Most common causes: Vehicle accidents (38%), falls (31%), violence (14%), sports (8%)
Current Treatments vs. Polylaminine
| Treatment | What It Does | Limitations |
|---|---|---|
| Stabilization surgery | Fixes the spine and decompresses the cord | Does not regenerate damaged tissue |
| Methylprednisolone | Anti-inflammatory corticosteroid | Controversial benefit, serious side effects |
| Intensive physiotherapy | Maintains muscle tone and mobility | Does not restore lost neural connections |
| Epidural electrical stimulation | Activates circuits below the injury | Requires implant; does not regenerate axons |
| Stem cells | Attempts to replace lost neurons | Inconsistent results; tumor risk |
| Robotic exoskeletons | Enables walking with technological support | Prohibitive cost; does not cure the injury |
What makes Polylaminine fundamentally different from all these approaches is that it attacks the problem at its root: instead of compensating for the loss of function or trying to replace destroyed neurons, it creates the conditions for the body itself to regenerate the damaged neural pathways.
Future Perspectives: What to Expect
Short-Term (2026-2028)
- Publication of complete data from primate tests
- Start of first human clinical trials (Phase I) in Brazil and potentially the USA
- Expansion of international collaborations
Medium-Term (2028-2032)
- Results of Phase I and II clinical trials
- Potential combination with complementary therapies (electrical stimulation, stem cells)
- Development of personalized protocols based on injury type and timing
Long-Term (2032+)
- If trials are successful, regulatory approval and start of clinical use
- More than 250,000 patients/year could benefit globally
- Economic impact: potential reduction of hundreds of billions in healthcare costs
- Expansion to other central nervous system regenerative applications
Combined Therapies: The Future Is Multifactorial
Most experts agree that the definitive treatment for spinal cord injuries will likely involve a combination of approaches, not a single solution. Polylaminine, as a bridge and regeneration stimulant, could be a central component of combined protocols:
- Polylaminine + Epidural Electrical Stimulation: Polylaminine regenerates axons; electrical stimulation activates newly formed circuits
- Polylaminine + iPSC Stem Cells: iPSCs provide new neurons; Polylaminine guides their connections
- Polylaminine + Gene Therapy: Genes encoding neurotrophic factors are inserted at the injury site to create an even more favorable environment
- Polylaminine + Intensive Rehabilitation: Specific motor exercises reinforce and refine regenerated connections
Conclusion: A New Era in Regenerative Medicine
The journey of Polylaminine — from an idea in a São Paulo laboratory to a potentially transformative therapy studied across 4 continents — is a testament to scientific perseverance and the human capacity to tackle problems that seemed unsolvable.
The path to medical offices is still long. Clinical trials may fail. Unforeseen obstacles may arise. But the data so far is, in the words of American neuroscientist Jerry Silver (Case Western Reserve University), "the most convincing I've seen in four decades of central nervous system regeneration research."
If Polylaminine fulfills its promise, we will be witnessing one of the greatest medical achievements of the 21st century — and it will have been born in Brazil.
Sources and References
- Hospital das Clínicas da USP — Institute of Orthopedics — Polylaminine development center
- World Health Organization (WHO) — Global data on spinal cord injuries
- National Institutes of Health (NIH) — Neural regeneration research
- Nature Neuroscience — Publications on spinal cord regeneration mechanisms
- The Lancet Neurology — Reviews on regenerative therapies in development





