H5N1 Bird Flu in Japan: Hokkaido Outbreak Forces Culling of 190,000 Chickens and Raises Global Alert
Date: March 6, 2026
Reading time: 22 minutes
Emoji: 🐔
On March 6, 2026, Japanese authorities confirmed the 21st outbreak of highly pathogenic avian influenza this season — this time at a farm housing 190,000 chickens in the town of Abira, Hokkaido. It is the fourth case on Japan's northernmost island, and every single bird will be culled, incinerated, and buried. As teams in biosecurity suits work through snow and sub-zero temperatures, the question echoing among scientists and health authorities worldwide is a singular one: is the H5N1 virus getting closer to becoming a real threat to human beings?
The Hokkaido Outbreak: Timeline and Response
The poultry farm in Abira, located in southern Hokkaido, reported abnormal signs in its flock on March 4, 2026. Workers noticed an unusual spike in chicken mortality — one of the classic early signs of highly pathogenic avian influenza (HPAI). Local veterinary authorities were immediately notified and collected samples for analysis.
Sequence of Events
| Date | Event |
|---|---|
| March 4 | Farm reports abnormal chicken mortality |
| March 4 (evening) | Rapid tests return positive for avian influenza |
| March 5 | Genetic testing confirms highly pathogenic H5N1 virus |
| March 6 | Government announces complete culling of all 190,000 birds |
| March 6-8 (est.) | Culling, incineration, and sanitary burial operations |
The results left no room for doubt: rapid antigen tests returned positive on the evening of March 4, and genetic analyses conducted by Japan's Ministry of Agriculture, Forestry and Fisheries (MAFF) confirmed the H5N1 subtype by the morning of March 5. Consequently, all 190,000 chickens at the farm were condemned to preventive culling — one of the most effective measures to prevent viral spread to other properties and, potentially, to regional wildlife.

Culling operations involve hundreds of workers equipped with full personal protective equipment — including waterproof coveralls, N95 masks, eye protection, and decontaminable boots. In Hokkaido, the work is even more challenging: temperatures can drop below -10°C (14°F), and accumulated snow hinders access to facilities and the transportation of biological waste.
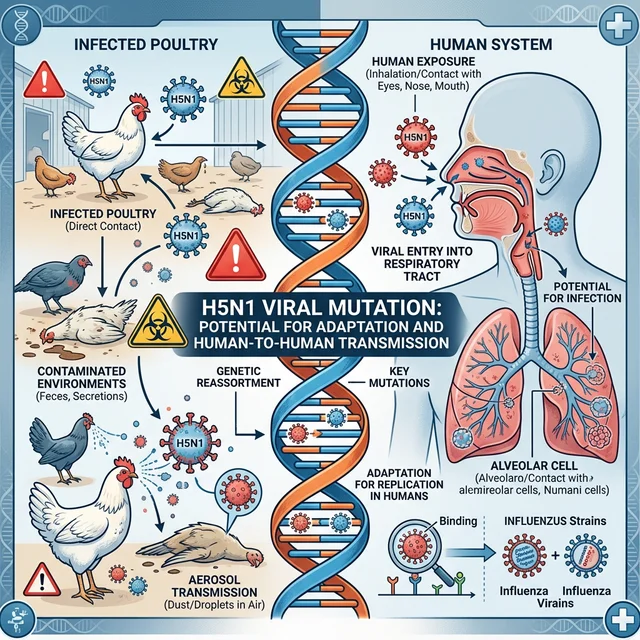
H5N1: A Virus in Constant Mutation
The H5N1 avian influenza virus is not new. It was first identified in geese in Guangdong province, China, in 1996. However, the variant currently circulating — belonging to clade 2.3.4.4b — represents a concerning evolution of the original pathogen, with the ability to infect a much broader range of species.
The Evolution of Clade 2.3.4.4b
Since 2020, the H5N1 clade 2.3.4.4b has been responsible for the largest wave of avian influenza ever recorded in history. The virus has spread across all inhabited continents, causing the death of hundreds of millions of domestic and wild birds and producing economic losses estimated in the tens of billions of dollars globally.

| Period | Most Affected Region | Species Impacted | Birds Culled |
|---|---|---|---|
| 2020-2021 | Europe and Central Asia | Domestic poultry, swans | ~50 million |
| 2021-2022 | North America | Domestic poultry, eagles | ~80 million |
| 2022-2023 | South America | Pelicans, sea lions | ~30 million |
| 2023-2024 | Antarctica and Oceania | Penguins, petrels | ~10 million |
| 2024-2026 | Global (Asia focus) | Birds, cattle, felines | Ongoing |
What makes clade 2.3.4.4b particularly alarming is its ability to jump between species with relative ease. Unlike previous variants, this H5N1 has already been detected in marine mammals (sea lions in Peru and Chile), dairy cattle (in the United States), domestic cats, and even polar bears in Arctic Canada. Each species jump represents an opportunity for the virus to accumulate mutations that could — in theory — make it better adapted for human-to-human transmission.
The Japanese Scenario: 21 Outbreaks in One Season
Japan's avian influenza season typically runs from autumn (October-November) through spring (March-April), coinciding with the migration of wild birds that carry the virus from breeding grounds in Siberia and Mongolia to the warmer lands of East and Southeast Asia.
Outbreak Distribution in the 2025-2026 Season
The Abira outbreak marks the 21st confirmed case of the 2025-2026 season and the 4th in Hokkaido. Japan's northernmost island has been particularly vulnerable as it is one of the first stopovers for migratory birds arriving from the Asian continent.
Japanese authorities follow a rigorous protocol, developed and refined over more than two decades of experience with HPAI outbreaks:
- Restriction zone (3 km): Total prohibition of bird and poultry product movement around the infected farm
- Surveillance zone (10 km): Intensified monitoring of all farms, with mandatory sample collection
- Complete preventive culling: Every bird at the infected farm is sacrificed, regardless of whether they show symptoms
- Full decontamination: Facilities are treated with specific disinfectants and inspected before allowing reintroduction of birds
- Contact tracing: Vehicles, equipment, and personnel who had contact with the farm are identified and monitored
The cost of these operations is astronomical. This season alone, Japan has already spent billions of yen on culling operations, producer compensations, and intensified biosecurity measures. Japan's poultry industry, which produces approximately 2.5 million tons of chicken meat and 2.6 billion eggs annually, faces mounting pressure on prices and availability.
The Risk to Humans: Where We Stand in 2026
The question that most concerns the global scientific and health community is not whether H5N1 can infect humans — it can and already has — but whether it is capable of developing the ability to transmit efficiently from person to person.
Confirmed Human Cases Globally
Since its emergence, H5N1 has infected more than 900 people worldwide, with a frighteningly high mortality rate of between 50% and 60%. However, it is important to contextualize these numbers:
- The vast majority of cases occurred in people with direct and prolonged contact with infected birds or contaminated environments (farms, live bird markets)
- Sustained human-to-human transmission has never been confirmed
- Recent cases (2024-2026) include cattle farmers in Texas and California who experienced mild conjunctivitis after contact with infected cattle — a far less severe presentation than classic cases
What Scientists Are Monitoring
The scientific community closely watches key mutations that could make H5N1 more dangerous to humans:
| Mutation | What It Means | Status in 2026 |
|---|---|---|
| PB2 E627K | Better replication in mammalian cells | Detected in some isolates |
| HA Q226L + G228S | Greater affinity for human receptors | Partially present |
| NA H275Y | Resistance to oseltamivir (Tamiflu) | Rare, but monitored |
| PA T271A | Greater virulence in mammals | Detected sporadically |
For H5N1 to become a human pandemic virus, it would likely need to accumulate multiple mutations simultaneously — a possible but statistically unlikely scenario in the short term, according to virologists. However, each new outbreak in mammals is a "molecular roulette," as virologist Ron Fouchier of Erasmus MC in the Netherlands has described it.
Economic Impact: Far Beyond the Farms
The effects of avian influenza transcend the poultry sector by a wide margin. The ongoing outbreak is creating ripple effects across multiple industries and economic sectors globally.
Agriculture and Food
Egg prices in international markets have hit historic records. In the United States, the average price of a dozen eggs has exceeded $7 in February 2026 — more than triple the price from two years earlier. In Japan, where eggs are a fundamental culinary ingredient (used in everything from ramen to tamago sushi), shortages are already affecting restaurants and food processing industries.
International Trade
Dozens of countries impose import restrictions on poultry products from affected nations. Japan, which normally exports part of its high-quality processed chicken production, faces barriers in Asian markets such as Hong Kong, Singapore, and Taiwan, which have suspended imports from regions with confirmed outbreaks.
Public Health Preparedness
Governments around the world are investing billions in preparation for a potential bird flu pandemic. The United States has already purchased millions of doses of human H5N1 vaccines, while the European Union has accelerated its development programs for universal influenza vaccines.
Prevention and Biosecurity Measures
Preventing avian influenza outbreaks depends fundamentally on rigorous biosecurity practices at poultry operations and effective monitoring of wild bird populations.

For Poultry Producers
- Complete isolation of facilities with physical barriers against wild birds (screens, covers, sonic scarecrows)
- Strict access control with registration of all visitors, vehicles, and equipment
- Decontamination procedures at all entrances and exits, including boot baths, vehicle washes, and clothing changes
- Daily monitoring of mortality and clinical signs, with mandatory reporting to veterinary authorities
- Avoid contact with wild birds, their habitats, and bodies of water frequented by migratory species
For the General Public
- Do not touch dead or visibly ill wild birds
- Report findings of large numbers of dead birds to local animal health authorities
- Cook poultry meat and eggs thoroughly (the virus is destroyed at temperatures above 70°C / 158°F)
- Wash hands frequently when visiting live bird markets or farms
- Stay informed about outbreaks in your region and follow health authority guidelines
Poultry Vaccination: The Debate
Some countries, such as China and Egypt, have adopted mass vaccination of domestic poultry against bird flu. Others, including Japan, the United States, and most European countries, prefer the "stamping out" strategy (complete culling and decontamination), arguing that vaccination can mask viral circulation and complicate epidemiological surveillance.
A growing international debate is whether this position can be sustained given the unprecedented scale of current outbreaks. France, after losing more than 20 million birds in successive outbreaks, began a pilot duck vaccination program in 2023 — a controversial decision being closely monitored by the international scientific community.
Future Outlook: What to Expect in 2026 and Beyond
The avian influenza outlook for the coming years remains uncertain, but there is scientific consensus on several fundamental points:
Possible Scenarios
- Optimistic scenario: Outbreaks decrease with the end of the migratory season (April-May), and the virus remains primarily confined to birds without significant adaptation to mammals
- Moderate scenario: H5N1 continues circulating endemically in domestic and wild bird populations, with sporadic cases in mammals and rare mild human infections
- Pessimistic scenario: The virus accumulates mutations in mammalian reservoirs (cattle, swine) that make it capable of efficient human-to-human transmission, triggering a pandemic
The World Health Organization (WHO) maintains H5N1 as one of the pathogens with the greatest pandemic potential on its priority watchlist. Meanwhile, governments and research institutions worldwide continue investing in genomic surveillance, vaccine and antiviral therapy development, and strengthening of rapid response systems.
Conclusion: Vigilance Is the Best Defense
The H5N1 outbreak in Hokkaido is yet another reminder that avian influenza remains one of the most significant health threats of our time. Although the probability of a human pandemic caused by H5N1 continues to be considered low to moderate, the potential consequences are so severe that they justify a continuous state of alertness and preparedness.
Japan's rapid and efficient response — from the farm's initial alert to the start of culling in less than 48 hours — demonstrates the importance of well-established protocols and a robust health surveillance culture. It is a model that other countries would do well to study and, wherever possible, adopt.
For ordinary citizens, the message is clear: stay informed, follow local health authority recommendations, and remember that biosecurity begins with simple actions — like avoiding contact with wild birds and ensuring proper cooking of poultry products.
Sources and References

- Japan Ministry of Agriculture, Forestry and Fisheries (MAFF) — Official HPAI outbreak communications
- World Health Organization (WHO) — Reports on human-animal influenza interface
- World Organisation for Animal Health (WOAH) — Global avian influenza outbreak database
- CDC — Centers for Disease Control (USA) — Human risk information and prevention
- Nature — Clade 2.3.4.4b Monitoring — Scientific publications on viral evolution
- The Statesman — Hokkaido outbreak coverage, March 2026





