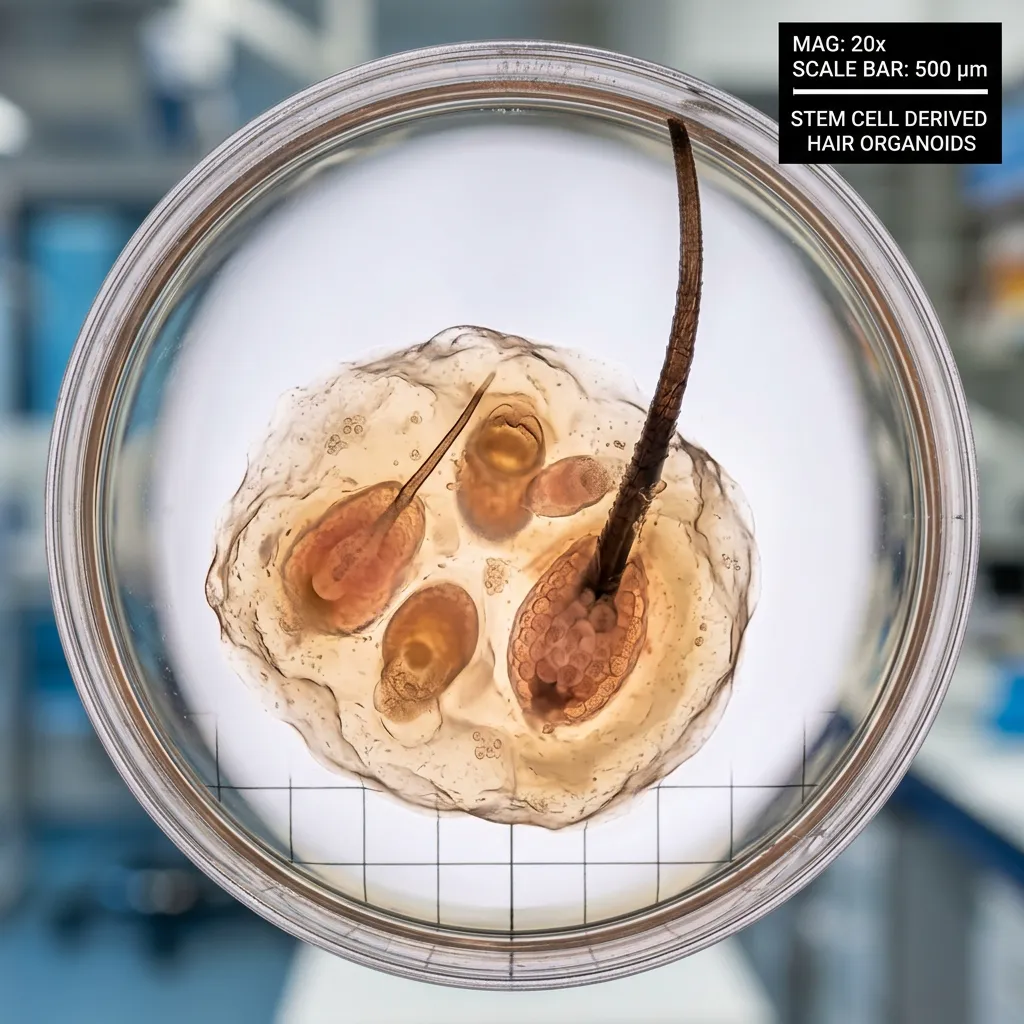
In a laboratory at Yokohama National University in Japan, a team of researchers has just accomplished what dermatology has been chasing for over four decades: growing complete, functional hair follicles from stem cells in a laboratory environment. Not simplified structures. Not organoids that vaguely resemble follicles under the microscope. Complete follicles — with sebaceous glands, arrector pili muscles, melanocytes producing natural pigment, and, most importantly, the ability to produce real hair shafts with the texture, color, and growth cycle indistinguishable from those that grow naturally on a human scalp.
The study, published in Science Advances in March 2026, represents what Dr. Tatsuto Kageyama, lead author, described as "the culmination of 30 years of collective effort across the field of hair biology." The significance goes far beyond cosmetics: this is a proof that complex mini-organs can be manufactured from scratch using human cells — a principle with implications that extend to organ regeneration, wound healing, and our fundamental understanding of how tissues form during embryonic development.
Why Growing a Hair Follicle Is So Incredibly Hard
To appreciate why this breakthrough took decades, you need to understand that a hair follicle is not a simple structure. It is one of the most complex mini-organs in the human body — a self-renewing machine that undergoes cycles of growth (anagen), regression (catagen), and rest (telogen) throughout a person's entire life. A single follicle contains at least 20 distinct cell types working in precise coordination.
The follicle requires an intricate dialogue between two fundamentally different tissue types: the epithelium (the outer skin layer) and the mesenchyme (the deeper connective tissue). This dialogue — known as epithelial-mesenchymal interaction — is what determines whether a follicle forms, what type of hair it produces, and when it cycles between growth phases. Replicating this interaction outside the body has been the holy grail of hair biology.
Previous attempts vs. the 2026 breakthrough:
| Approach | Year | Result | Limitation |
|---|---|---|---|
| Dermal papilla cell culture | 1984-2020 | Cells lose inductive capacity in culture | Cannot form complete follicles |
| Embryonic skin grafts (mice) | 2010 | Complete follicles | Requires embryonic tissue, not scalable |
| 3D spheroid cultures | 2015 | Partial follicle structures | No hair shaft production |
| iPSC-derived organoids | 2020 | Small follicle-like structures | Low efficiency, inconsistent |
| Yokohama method (hydrogel scaffold) | 2026 | Complete functional follicles | First lab-grown full follicle |
The Yokohama Method: How They Did It
The key innovation was a custom-designed collagen hydrogel scaffold — a three-dimensional biomaterial that mimics the extracellular matrix surrounding developing follicles in the embryo. This scaffold provides the mechanical signals, the spatial organization, and the biochemical gradients that guide stem cells to self-organize into follicular structures.
The three-step process:
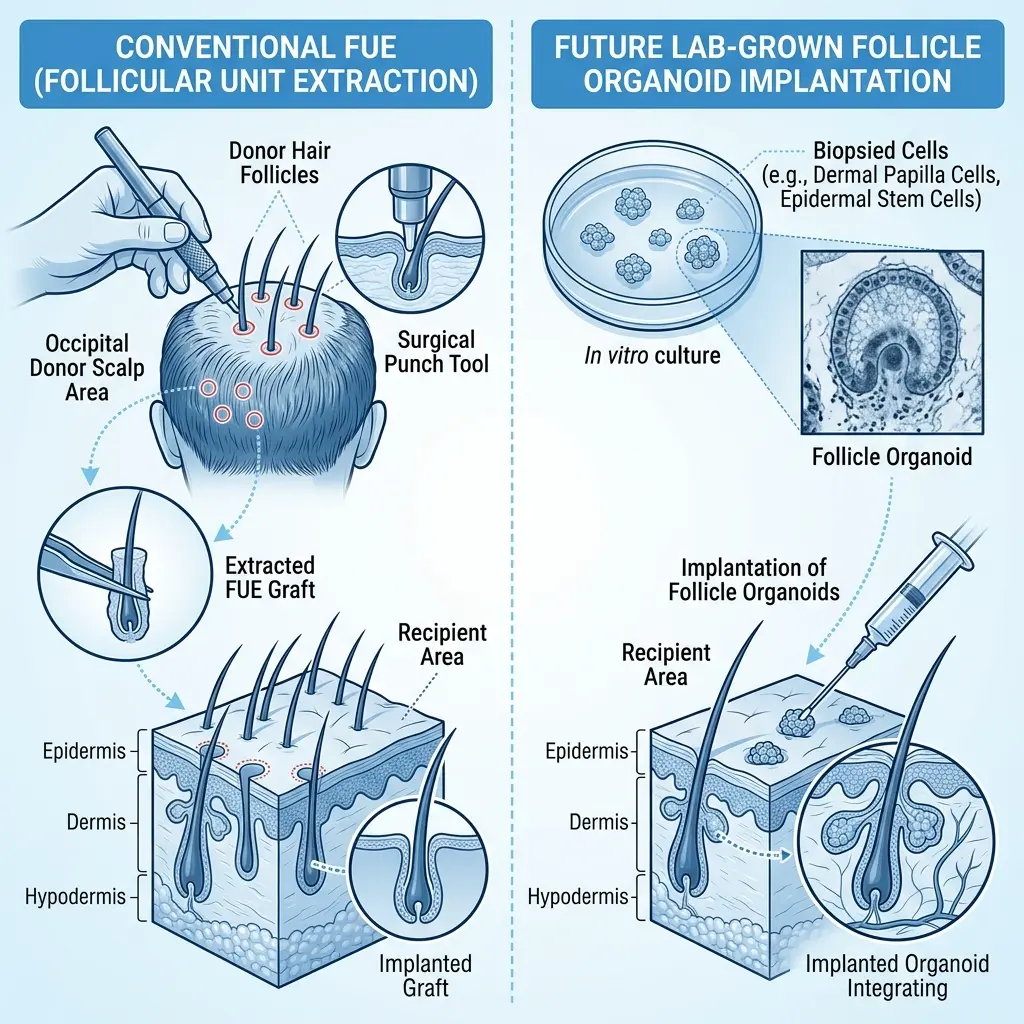
Step 1 — Cell sourcing: The team isolated two cell populations from small skin biopsies: epidermal stem cells (from the outer skin layer) and dermal papilla cells (the mesenchymal component that controls hair growth). Both can be obtained from a tiny punch biopsy — roughly 3mm in diameter — taken from the back of a patient's scalp.
Step 2 — Scaffold assembly: The cells were seeded into the collagen hydrogel at precisely controlled densities and ratios. The ratio of epithelial to mesenchymal cells proved critical: a 3:1 ratio produced the most consistent results. The hydrogel's stiffness was tuned to match the mechanical properties of embryonic skin — softer than adult skin, which turns out to be essential for follicle initiation.
Step 3 — Culture and maturation: Over 23 days in optimized conditions (with specific growth factors and signaling molecules), the cells self-organized into complete follicular structures. By day 23, hair shafts were visibly emerging from the organoids, growing at approximately 0.3mm per day — consistent with the growth rate of natural human hair.
The results were remarkable: 96% of organoids produced at least one functional follicle, and each organoid contained an average of 4.2 follicles — enough to provide meaningful coverage when transplanted into nude mice (which lack their own hair, providing a clean substrate for testing).
What This Means for the 800 Million People with Hair Loss
The global hair loss market is valued at approximately US$ 9.5 billion in 2026 and is projected to reach US$ 15 billion by 2032. Currently, the available options are:
- Minoxidil (Rogaine): Slows loss, does not restore hair. Works in about 40% of users. Must be used indefinitely.
- Finasteride (Propecia): Blocks DHT hormone. Effective but carries side effects including reduced libido in 1-3% of users.
- PRP therapy (Platelet-Rich Plasma): Emerging, evidence mixed, expensive ($500-2,000 per session), requires repeated treatments.
- Hair transplant surgery (FUE/FUT): The gold standard for restoration, but limited by donor area — you're moving existing follicles, not creating new ones. Costs $4,000-$30,000 depending on coverage needed.
The Yokohama method removes the most fundamental limitation of hair transplants: the finite donor supply. From a single 3mm biopsy, the team estimated that enough cells could be cultured to produce 10,000+ follicles — far exceeding the 2,000-4,000 grafts typical of a modern FUE procedure. This means complete scalp coverage could theoretically be achieved regardless of how advanced the hair loss is.
Hair loss by the numbers in 2026:
| Statistic | Data |
|---|---|
| Men affected by androgenetic alopecia globally | ~800 million |
| Women affected by hair thinning | ~350 million |
| Average age of onset (men) | 25-35 years |
| Global hair transplant market | US$ 9.5 billion |
| Average cost of FUE transplant | US$ 8,000-15,000 |
| Grafts available from donor area | 4,000-6,000 (lifetime max) |
| Grafts possible from single biopsy (Yokohama method) | 10,000+ |
The Road to Human Trials
The Yokohama team has already begun preliminary discussions with Japan's Pharmaceuticals and Medical Devices Agency (PMDA) about the regulatory pathway to human trials. Dr. Kageyama estimates that Phase I clinical trials could begin by 2028-2029, targeting patients with androgenetic alopecia (the most common form of male-pattern and female-pattern hair loss).
The challenges remaining before clinical application include:
1. Scalability and manufacturing
Current production is done in small laboratory batches. Scaling to produce tens of thousands of follicles per patient while maintaining quality and consistency will require significant bioprocess engineering investment.
2. Vascularization
Lab-grown follicles need to connect to the blood supply after implantation. Initial mouse experiments showed successful vascularization within 10-14 days, but human skin is thicker and more complex, requiring further optimization.
3. Color and texture matching
While the follicles produce pigmented hair, achieving precise color and curl pattern matching across the entire scalp remains a technical challenge. The team is developing methods to sort and select follicles based on melanocyte activity before implantation.
4. Long-term cycling
Natural follicles cycle through growth phases for a lifetime. Demonstrating that lab-grown follicles maintain this cycling behavior over years (not just months) will be critical for regulatory approval.
The Bigger Picture: Beyond Hair
What makes this breakthrough truly significant extends far beyond baldness treatment. The principles demonstrated — growing a complete, functional mini-organ from adult stem cells using a biomimetic scaffold — have implications across regenerative medicine:
Skin wounds: The same follicular organoids could be transplanted into burn scars or chronic wound sites, promoting healing with hair-bearing skin rather than the hairless scar tissue that currently results from severe burns.
Sweat glands: Researchers at Fudan University in China are already adapting similar hydrogel approaches to grow sweat gland organoids, which could benefit burn patients who lose thermoregulation capacity.
Drug testing: Hair follicles are one of the first organs affected by chemotherapy. Lab-grown follicles could serve as testing platforms to develop cancer drugs that spare hair — addressing the significant psychological burden of chemotherapy-induced alopecia.
Understanding aging: Hair graying and thinning are among the most visible signs of aging. Growing follicles in controlled conditions allows researchers to study these processes in unprecedented detail, potentially leading to anti-aging interventions that extend far beyond cosmetics.

The Psychological Weight of Hair Loss: Why This Matters Beyond Vanity
It's tempting to dismiss hair loss research as a cosmetic triviality — a vanity problem unworthy of serious scientific attention. This dismissal fundamentally misunderstands the psychological devastation that alopecia inflicts on hundreds of millions of people worldwide, and it ignores decades of robust clinical evidence documenting the mental health consequences of involuntary hair loss.
A comprehensive meta-analysis published in the British Journal of Dermatology in 2024, aggregating data from 47 studies and over 12,000 participants, found that individuals with significant hair loss exhibited:
- 45% higher rates of clinical anxiety compared to age-matched controls
- 38% higher rates of clinical depression
- 62% reported avoiding social situations due to self-consciousness about their appearance
- 28% of men under 30 with advanced hair loss reported it as their "primary source of psychological distress"
- Women with alopecia reported even greater psychological impact, with 71% describing their hair loss as "devastating" to their self-image
The psychological burden is particularly acute in younger patients. Dr. Amanda Rodrigues, a clinical psychologist specializing in appearance-related distress at University College London, explains: "We live in a culture where hair is deeply intertwined with identity, attractiveness, and perceived health. When a 25-year-old man sees his hairline receding, he's not worried about hair — he's worried about being perceived as older, less attractive, and less vital. The psychological distress is as real and measurable as any medical condition."
For women, the impact is often even more severe. Female hair loss — which affects approximately 40% of women by age 50 — carries an additional stigma because society normalizes male baldness but often treats female hair thinning as a sign of illness or aging that must be hidden. Cancer patients undergoing chemotherapy consistently rank hair loss among their most dreaded side effects — often above nausea and fatigue.
The economic costs are equally staggering. Americans alone spend US$ 3.5 billion annually on hair loss treatments, products, and procedures — much of it on solutions with minimal efficacy. The proliferation of unproven supplements, laser caps, and miracle serums targeting desperate consumers represents one of the largest wellness industry segments with the weakest evidence base. A truly effective, one-time regenerative treatment would not merely transform aesthetics — it would eliminate an entire ecosystem of exploitation built on hope and insecurity.
The Global Race: Who Else Is Working on Hair Regeneration
Yokohama's breakthrough doesn't exist in isolation. Multiple research groups worldwide are pursuing parallel strategies for hair follicle regeneration, creating a competitive landscape that could accelerate commercialization:
Key competitors in hair regeneration (2026):
| Organization | Approach | Status | Differentiator |
|---|---|---|---|
| dNovo/Columbia University (USA) | 3D-printed follicle molds with dermal papilla cells | Phase I trials in planning | Uses scaffolds shaped like natural follicles |
| RIKEN Center (Japan) | iPSC-derived hair follicle germs | Preclinical (mice) | Fully pluripotent source, unlimited expansion |
| Stemson Therapeutics (USA) | iPSC-derived dermal papilla cells + bioengineered scaffolds | Preclinical | Partnership with AbbVie, significant funding |
| HairClone (UK) | Dermal papilla cell banking and multiplication | Early clinical | Focus on banking young cells for future use |
| RepliCel Life Sciences (Canada) | Autologous dermal sheath cup cells | Phase II completed | RCH-01 injections for hair thickening |
| Yokohama Nat'l University (Japan) | Complete organoid with hydrogel scaffold | Proof of concept | First full functional follicle from scratch |
The investment landscape reflects growing confidence in the field. Stemson Therapeutics raised US$ 22.5 million in Series B funding in late 2025, backed by pharmaceutical giant AbbVie. dNovo closed a US$ 15 million seed round. The total venture capital invested in hair regeneration startups between 2023 and 2026 exceeds US$ 200 million — a figure that would have been unthinkable a decade ago when hair biology was considered a scientific backwater.
What distinguishes the Yokohama achievement from its competitors is completeness: while most approaches focus on one component of the follicle (dermal papilla cells, hair germ structures, or scaffolds alone), the Yokohama team produced a fully assembled, cycling, pigmented follicle — the first time anyone has done so from adult human cells outside the body. It's the difference between building a car engine and building an entire car that actually drives.
The convergence of multiple approaches also increases the probability that at least one pathway will reach clinical application within the decade. Even if Yokohama's specific method encounters scaling hurdles, the foundational principles it validated — hydrogel scaffolding, self-organization from defined cell ratios, growth factor timing — will inform every competitor's work. Scientific breakthroughs rarely benefit just their originators; they illuminate the entire field.
FAQ — Frequently Asked Questions
When will lab-grown hair follicle treatment be available?
Phase I clinical trials could begin by 2028-2029 in Japan. If successful, broader availability could follow by 2032-2035 in regulated markets. The timeline depends heavily on regulatory approval processes and manufacturing scalability.
How much would the treatment cost?
Early estimates suggest the initial cost could be comparable to current hair transplant surgery (US$ 8,000-20,000), but with significantly better coverage and results. As the technology matures and scales, costs are expected to decrease substantially.
Would the treatment work for all types of baldness?
The Yokohama method targets androgenetic alopecia (the most common type), but the principles could apply to alopecia areata (autoimmune) and scarring alopecias. Clinical trials will need to evaluate each condition separately.
Is this treatment for both men and women?
Yes. The research applies to both male-pattern and female-pattern hair loss, as the stem cells can be derived from any individual regardless of sex.
What are the risks?
Potential risks include infection at transplant sites, immune rejection of cell products, and theoretical (though undemonstrated) oncogenic potential from cultured stem cells. These will be rigorously evaluated in clinical trials.
Sources and References
- Science Advances: "Generation of Hair-Bearing Skin Organoids from Human Stem Cells" — March 2026
- Yokohama National University Press Release — March 2026
- Dr. Tatsuto Kageyama et al., Department of Biomedical Engineering, Yokohama National University
- International Society of Hair Restoration Surgery: 2026 Practice Census
- Global Hair Loss Treatment Market Report — Grand View Research, 2026
- Nature Reviews Drug Discovery: "Hair Follicle Regeneration: Progress and Prospects" — March 2026





