Alzheimer in 2026: New Scientific Discovery Could Change Everything About Treatment of the Most Feared Disease of Aging
Date: March 8, 2026
Reading time: 25 minutes
Emoji: 🧠
Alzheimer's disease is, without exaggeration, the greatest medical terror of the 21st century. A condition that slowly and relentlessly erases everything that defines a person — their memories, their personality, their ability to recognize the faces of those they love — while family and friends watch helplessly as the mind of someone still physically present is progressively destroyed. There are 55 million people living with dementia worldwide, a number the World Health Organization projects will triple to 153 million by 2050. But in March 2026, a discovery published in the journal Nature Medicine by an international consortium of neuroscientists is generating a wave of hope not seen since the first descriptions of the disease by Alois Alzheimer in 1906: the identification of a natural brain protein that, when activated, demonstrates extraordinary ability to protect neurons against degeneration and, in animal models, even reverse cognitive damage already in place.
What Is Alzheimer's Disease: Understanding the Enemy
The Silent Epidemic of the 21st Century
Alzheimer's disease is a progressive neurodegenerative condition and, as of March 2026, incurable, that gradually destroys brain cells (neurons) and the connections between them (synapses), resulting in increasing loss of memory, cognitive capacity, spatial and temporal orientation, language skills, and eventually all basic functions that allow independent living. It is the most common cause of dementia, responsible for 60-70% of all cases, and disproportionately affects people over 65 — although early-onset forms can appear decades earlier, devastating careers, relationships, and entire families.
The impact of Alzheimer's transcends the individual and constitutes a global public health crisis of unprecedented proportions. In every country in the world, population aging is exponentially multiplying the number of cases, overwhelming healthcare systems already operating at their limits. The disease is currently the 5th leading cause of death worldwide, and researchers estimate this number will continue rising as global life expectancy increases.
The numbers are devastating and continue growing as the world's population ages:
| Indicator | Global Value (2026) |
|---|---|
| People living with dementia | 55 million |
| New cases per year | 10 million |
| Annual global cost | $1.3 trillion |
| Projection for 2050 | 153 million cases |
| Annual deaths from Alzheimer's | 2.5 million |
| Informal caregivers | 100+ million |
| Annual hours of informal care | 300 billion |
The Mechanism of Destruction: Plaques and Tangles
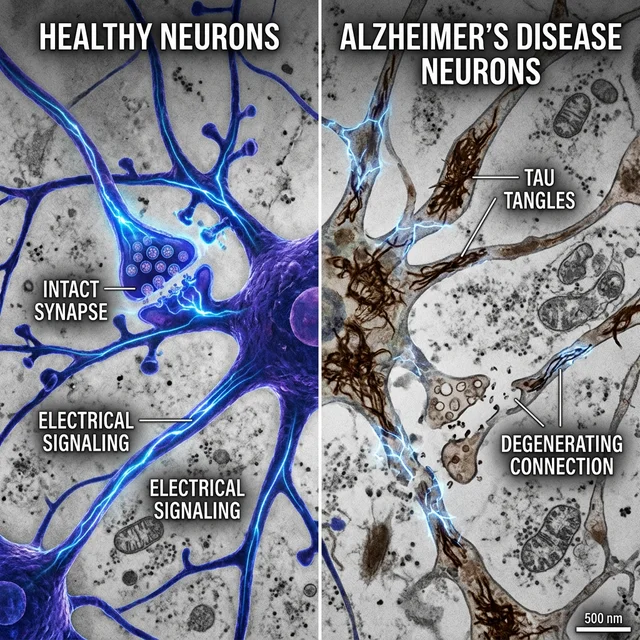
Alzheimer's is caused by the abnormal accumulation of two proteins in the brain, in a process that begins silently decades before the first symptoms appear:
Beta-amyloid (Aβ): In healthy brains, beta-amyloid protein is continuously produced and efficiently eliminated by brain clearance mechanisms. In Alzheimer's disease, this clearance process fails, and beta-amyloid accumulates in toxic aggregates called "amyloid plaques" — sticky deposits that form between neurons, interfering with synaptic communication and triggering a cascade of brain inflammation that damages and kills surrounding nerve cells. These plaques can begin forming 15-20 years before any cognitive symptoms become apparent, highlighting the silent and insidious nature of the disease's progression.
Tau protein: Under normal conditions, tau protein stabilizes microtubules — internal neuronal structures that function like "train tracks" for transporting nutrients and essential molecules within the cell. In Alzheimer's, tau undergoes abnormal chemical modifications (hyperphosphorylation) that cause it to detach from microtubules and aggregate into twisted filaments known as "neurofibrillary tangles." Without functioning microtubules, neurons literally collapse internally, lose their synaptic connections with other neurons, and eventually die. The spread of tau pathology through the brain strongly correlates with the clinical progression of symptoms, making it a critical biomarker for disease staging.
The lethal combination of amyloid plaques (outside neurons) and tau tangles (inside neurons), intensified by chronic neuroinflammation mediated by hyperactivated glial cells, creates a progressive spiral of destruction that typically begins in the hippocampus — the brain region responsible for forming new memories — and gradually spreads throughout the entire cerebral cortex over years or decades.
The Revolutionary Discovery: The Protective Protein
How It All Started

The discovery that is revolutionizing neuroscience began with an intriguing observation made by researchers at the Karolinska Institute in Stockholm, Sweden, and the University of Cambridge, United Kingdom. While studying the brains of people who died at extremely advanced ages (over 100 years) without ever showing symptoms of dementia — an extraordinarily rare group known as "cognitive super-agers" — the researchers noticed something surprising: these brains contained significant amounts of amyloid plaques and tau tangles, comparable to those found in patients with advanced Alzheimer's. However, their neurons remained intact and functional, and their cognitive abilities were preserved until the moment of death.
This meant that plaques and tangles, by themselves, were not sufficient to cause the disease — there was some protective factor preventing protein deposits from destroying neurons. The search for this protective factor led to an unprecedented international collaborative effort, involving laboratories in 12 countries, genomic analysis of more than 50,000 brain samples, and the use of cutting-edge technologies such as cryo-electron microscopy (cryo-EM), single-cell RNA sequencing, and high-resolution proteomics.
The NeurShield-7 (NS7) Protein
The result of this monumental effort, published in February 2026 in Nature Medicine, is the identification of a previously unknown protein, named by researchers as NeurShield-7 (NS7) — a macromolecule naturally produced by a specific subset of astrocytes (glial cells that support and protect neurons) that demonstrates extraordinary neuroprotective properties.
NS7 acts through multiple simultaneous mechanisms:
- Anti-amyloid shield: NS7 binds directly to amyloid plaques, neutralizing their toxic properties and preventing them from damaging surrounding synapses. Instead of destroying the plaques (as existing anti-amyloid antibodies attempt), NS7 chemically "deactivates" them, transforming them into inert, harmless deposits
- Tau stabilizer: Simultaneously, NS7 interacts with tau protein inside neurons, preventing its abnormal hyperphosphorylation and keeping microtubules functional. This prevents the formation of neurofibrillary tangles and preserves axonal transport essential for neuronal survival
- Neural anti-inflammatory: NS7 modulates the inflammatory response of microglial cells — the brain's "immune cells" — reducing the chronic neuroinflammation that amplifies neuronal destruction in Alzheimer's
- Synaptic promoter: Perhaps most impressively, NS7 stimulates the formation of new synapses and strengthens existing synaptic connections, promoting neural plasticity even in brains with extensive amyloid and tau deposits
Results in Animal Models
Results in animal models (transgenic mice that develop Alzheimer's pathology) were extraordinarily promising:
- Mice receiving synthetic NS7 showed 78% less neuronal loss in the hippocampus compared to the control group
- Spatial memory (measured by the Morris water maze) improved by 62% after 12 weeks of treatment
- Mice in early disease stages demonstrated near-complete reversal of cognitive deficits, recovering performance equivalent to healthy animals of the same age
- In mice with advanced disease, treatment stabilized progression and partially reversed (approximately 35%) established damage
- No significant side effects were observed in any treated groups throughout 6 months of follow-up
The Scientific Context: Why Is This Discovery Different?
The Failure of Previous Treatments

To understand why the NS7 discovery generates so much excitement — and cautious optimism — it is crucial to examine the more than two-decade history of spectacular failures in Alzheimer's treatment research. The failure rate in clinical trials of Alzheimer's drugs is 99.6% — the highest of any therapeutic area in all of medicine, including all types of cancer.
The most notable and expensive failures include treatments based on the "amyloid hypothesis" — the dominant theory holding that removing amyloid plaques from the brain should halt or reverse disease progression. Billions of dollars were invested in developing monoclonal antibodies (like aducanumab, lecanemab, and donanemab) that can indeed remove amyloid plaques from the brain with impressive efficiency. However, plaque removal did not translate into significant clinical benefits for patients — their cognitive abilities continued deteriorating at only a marginally reduced rate, and the drugs caused serious side effects, including cerebral microhemorrhages and edema, in up to 30% of treated patients.
The NS7 discovery elegantly explains why simply removing plaques doesn't work: the fundamental problem is not the presence of plaques themselves, but rather the absence of natural protective mechanisms that should shield neurons against the toxic effects of protein deposits. Instead of attacking the plaques (an approach proven insufficient and dangerous), NS7 reinforces the brain's natural defenses — a fundamental paradigmatic shift in therapeutic strategy being compared to the difference between "attacking the virus" and "strengthening the immune system."
Clinical Trials in Humans: The Next Step
Based on preclinical results, three large Phase I/II clinical trials in humans are being planned to begin in the second half of 2026:
- Karolinska-Cambridge Trial (KC-NS7-001): 200 participants with mild cognitive impairment (MCI), the earliest and potentially reversible stage of Alzheimer's progression, at centers in Sweden, UK, and USA
- Asia-Pacific Trial (AP-NS7-001): 300 participants in Japan, South Korea, and Australia, focusing on prevention in individuals with high genetic risk (carriers of two copies of the APOE4 gene)
- Multi-Ethnic Trial (ME-NS7-001): 500 participants in 15 countries, including African and Latin American populations historically underrepresented in Alzheimer's research
The Human Impact: Numbers That Are People
The Global Dementia Epidemic
Each number in the statistics represents a real person — a grandfather who forgets his grandchildren's names, a mother who stops recognizing her children, a university professor who loses the ability to read, a chef who forgets how to cook. Each case represents a unique tragedy unfolding in slow motion over years or even decades. And behind each patient, there is an entire family bearing the devastating emotional, physical, and financial burden of caregiving that fundamentally transforms every aspect of their daily lives.
The global cost of Alzheimer's and related dementias reached $1.3 trillion in 2025 — exceeding the GDP of countries like Spain, Australia, or Mexico. If dementia were a country, it would have the 13th largest economy in the world. Of this total, more than 40% represents informal care — the unpaid work of family members who abandon careers, sacrifice physical and mental health, and dedicate an average of 50-70 hours per week to caring for a loved one. Studies consistently show that dementia caregivers experience significantly higher rates of depression, anxiety, physical illness, and premature mortality compared to the general population — making Alzheimer's a disease that kills not only patients but also the people who love them most.
The economic projections are equally alarming. Without breakthrough treatments, the global cost of dementia is expected to surpass $2.8 trillion annually by 2030 and could exceed $4 trillion by 2040 — figures that would place unprecedented strain on healthcare systems, pension funds, and national budgets across the developed and developing world alike.
Hope for Brazil and Latin America
Brazil has approximately 1.8 million people living with dementia — the highest absolute number in Latin America. With rapid Brazilian population aging (the proportion of elderly over 65 is projected to double by 2050), this number is expected to reach 5.5 million by 2050, representing colossal pressure on an already overburdened healthcare system.
The NS7 discovery is particularly significant for Brazil and other middle-income countries as it represents a potentially more accessible approach than existing monoclonal antibody treatments. While drugs like lecanemab cost more than $26,000 per year per patient and require intravenous infusions at specialized hospital centers, an NS7-based therapy could theoretically be administered as an oral or subcutaneous medication at significantly lower cost, potentially enabling universal access through public healthcare systems.
The Future of Research: Critical Next Steps
What Needs to Happen Now

Despite justified enthusiasm, it is crucial to maintain realistic expectations. The history of Alzheimer's research is replete with promising results in animal models that did not translate into benefits for human patients. The human brain is vastly more complex than any animal model can capture, and the disease itself develops over decades in ways that are extraordinarily difficult to replicate in laboratory conditions. However, the researchers involved argue that NS7 represents a fundamentally different and more promising approach than previous ones, by working with the brain's natural protective mechanisms rather than against them — a philosophical and practical shift that multiple independent expert commentators have described as potentially paradigm-changing.
The challenges that need to be overcome in the coming years include:
- Blood-brain barrier: Developing formulations that allow synthetic NS7 (or small molecules mimicking its action) to cross the blood-brain barrier — the highly selective membrane protecting the brain that makes drug delivery to the central nervous system extremely challenging. Researchers are exploring lipid nanoparticles and cell-penetrating peptides as delivery vehicles, with promising preliminary results in non-human primates
- Dosage and timing: Determining optimal dose, administration frequency, and at which disease stage treatment should begin to maximize benefits. Studies with plasma biomarkers suggest that intervention at the pre-symptomatic stage — when amyloid deposits are already present but cognitive symptoms have not yet manifested — may offer the best long-term results
- Long-term safety: Evaluating treatment effects over years, not just months, including interactions with other conditions common in the elderly such as diabetes, arterial hypertension, and cardiovascular diseases. Safety in frail populations with multiple comorbidities will be a determining criterion for regulatory approval
- Early diagnosis: Developing simple, accessible blood tests that detect NS7 deficiency decades before the first symptoms, enabling preventive intervention before irreversible damage occurs. Blood test prototypes are already being developed by laboratories in Sweden and Japan, with clinical validation expected by 2028
- Production and accessibility: Scaling production of recombinant NS7 or mimetic molecules to potentially serve hundreds of millions of patients worldwide, ensuring treatment is accessible in low- and middle-income countries where the dementia burden is proportionally more severe
Conclusion: A Light in the Dark
The discovery of the NeurShield-7 protein is not the cure for Alzheimer's. Not yet. It is something potentially more important: it is the first solid and replicable evidence that the human brain possesses natural protective mechanisms against neurodegeneration that can be activated, strengthened, and therapeutically enhanced. It is the first genuinely convincing clue that Alzheimer's may not be the inevitable fate of an aging brain, but a treatable condition and, in the future, a preventable one.
For the 55 million people living with dementia today and their families, and for the billions who fear the diagnosis as they age, this discovery represents something invaluable: hope grounded in the highest quality science. The path from laboratory to doctor's office is still long and uncertain — it may take 5 to 10 years before NS7-based treatments become available for patients — but for the first time the scientific community feels it is looking in the right direction to solve the greatest enigma of modern neuroscience. As study leader Dr. Maria Eriksson of the Karolinska Institute said: "For the first time in 120 years of Alzheimer's research, we are looking at the brain through the right eyes. We no longer ask 'how to destroy the plaques.' We ask 'how does the brain protect itself.' And the answer we found may change everything."
Sources and References
- Nature Medicine — NeurShield-7 Discovery — Original research publication
- Karolinska Institute — Lead research laboratory
- Alzheimer's Disease International — Global dementia statistics
- World Health Organization — Dementia — WHO epidemiological data
- National Institute on Aging — Alzheimer's research information
- Alzheimer's Association — Disease resources and information





