Digital Twins: AI-Created Organ Replicas That Could Revolutionize Medicine
Imagine that, before taking a medication, your doctor could first test it on a virtual copy identical to YOUR liver, YOUR lungs, YOUR kidneys — checking if it works, if it causes side effects, and what the ideal dose is for YOUR body specifically. Not on a lab rat. Not on a group of 1,000 volunteers whose bodies may be radically different from yours. On your digital twin. It sounds like science fiction, but it's exactly what three of the world's most prestigious institutions — Imperial College London, the University of Oxford, and pharmaceutical giant GSK — are building right now, in 2026.
What Are Digital Twins

From Jet Engines to the Human Body
The concept of a "digital twin" isn't new — it was born in engineering. NASA developed the first digital twins in the 1960s to simulate Apollo spacecraft conditions. Rolls-Royce uses digital twins of their jet engines to predict failures before they happen. BMW simulates entire factories digitally before building a single part.
The central idea is simple: create a virtual replica so precise of a real object that you can simulate scenarios on the digital model and trust that the results apply to the real world.
The 2026 revolution is applying this logic to the human body — specifically, to individual organs.
The Imperial-Oxford-GSK Project
| Organ | Why this one first | Status |
|---|---|---|
| Lung | Respiratory diseases are the 3rd cause of death globally; COVID-19 accelerated urgency | Functional prototype |
| Liver | 50% of drug failures in clinical trials are due to hepatotoxicity | In development |
| Kidney | Chronic kidney disease affects 850 million people; nephrotoxic drugs are common | In development |
How It Works

The Architecture of a Digital Organ Twin
Layer 1: Biological Data — Medical images, genomic sequencing, blood tests, clinical history
Layer 2: Computational Models — Fluid dynamics, mechanical models, biochemical models, cellular models
Layer 3: Artificial Intelligence — Neural networks trained on millions of anonymized medical records, generative models, reinforcement learning, GPU cloud processing
The result is a 3D interactive model of a specific organ of a specific patient that behaves — in terms of physiology, metabolism, and substance response — the same way as the real organ.
Applications: What Changes in Practice
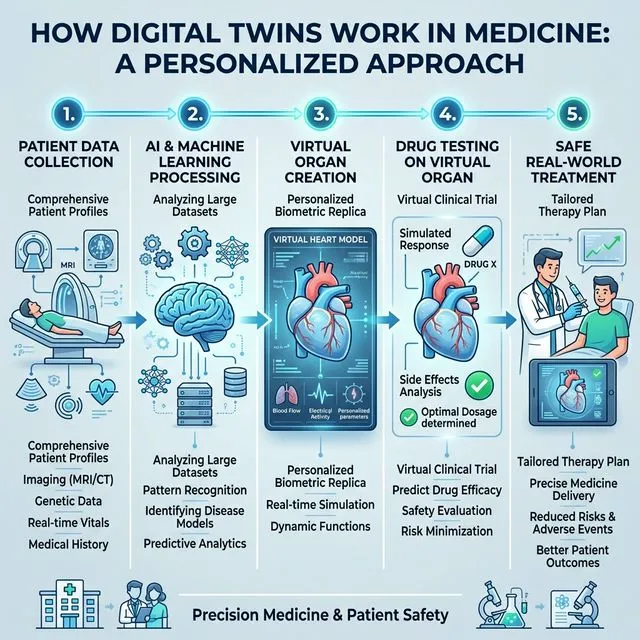
1. Drug Testing Without Guinea Pigs
New drug development takes 10-15 years and costs $2.6 billion on average. With digital twins:
- Test drugs on thousands of digital twins before touching a human
- 50% of drugs fail in Phase II due to liver toxicity — testable digitally
- Simulations that take months in vivo can take hours in silico
2. Real Personalized Medicine
With digital twins, personalized medicine means simulating YOUR body's response to medication before prescribing it.
3. Predicting Diseases Before Symptoms
A continuously updated digital twin could detect subtle organ degradation months or years before symptoms appear.
Challenges and Limitations
Privacy: Who Has Access to Your Twin?
A digital organ twin contains the most intimate medical information possible. The project adopts homomorphic encryption, layered anonymization, and GDPR/NHS compliance.
Accuracy: How Far Does Simulation Reflect Reality?
The human body has 37 trillion cells. Replicating this complexity completely is computationally impossible today. 2026's digital twins are sophisticated approximations — good enough to predict drug toxicity, not good enough to simulate cancer emergence.
Equity: Two-Speed Medicine?
If digital twins become standard, who benefits first? Probably patients in wealthy countries — amplifying global health inequality.
The Horizon: 2030 and Beyond
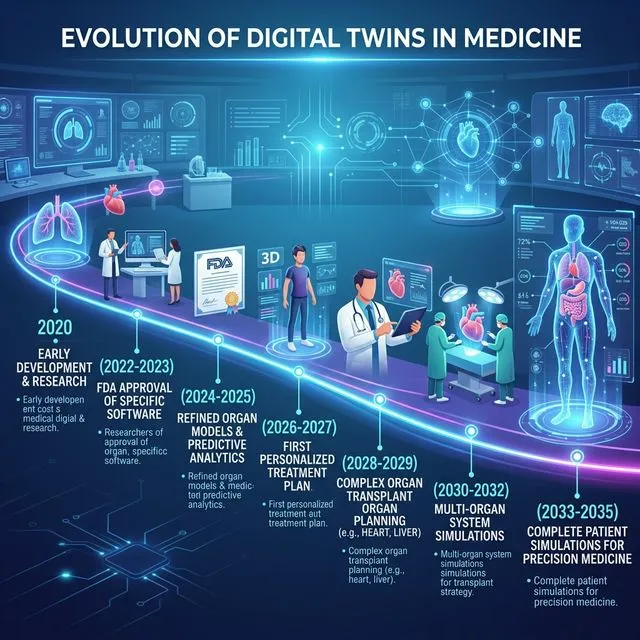
By 2030:
- Digital twins of 5 major organs will be available for clinical use
- 50% of Phase I trials will be conducted totally or partially in silico
- Drug development costs will drop 30-40%
- Time to approval will reduce from 10-15 to 5-8 years
The Global Digital Twins Race
- Siemens Healthineers: €300 million in "Digital Twin Heart"
- Dassault Systèmes: "Living Heart Project" used by FDA
- Microsoft + Novartis: Azure cloud for industrial-scale simulations
- China: ¥15 billion ($2.1 billion) investment announced in 2025
The global health digital twins market: $1.8 billion in 2025, projected $12.5 billion by 2030.
The Ethical Debate: End of Animal Testing?
Digital organ twins carry a promise beyond medical efficiency — potentially reducing or eliminating animal testing. Currently, 100-200 million animals are used annually in preclinical tests. Over 90% of drugs that work in animals fail in humans. Digital twins based on real human data eliminate the interspecies translation problem.
The FDA Modernization Act 2.0 (2023) removed the requirement for animal testing as a prerequisite for human clinical trials, opening the door for digital twin alternatives.
What It Means for Brazil
SUS and Personalized Medicine
The SUS (Brazilian Unified Health System) serves over 190 million Brazilians. Challenges include data infrastructure (many units still use paper records), computational costs, and medical training in AI. Opportunities include Brazil's genetically diverse population producing more representative digital twins.
Brazilian research through USP, UNICAMP, UFMG, and the GAIA project (FAPESP) is developing drug response simulation models based on Brazilian genomic data. ANVISA has signaled it may accept computational simulation data as complementary evidence in drug registration processes.
Frequently Asked Questions (FAQ)
How long until I can have a digital twin of my body?
Single-organ models should be available for limited clinical use by 2030. Full-body digital twins (interconnecting lung, liver, kidney, heart, and brain) are projected for 2035-2040.
Can digital twins completely replace animal testing?
Not completely in the medium term — but they can drastically reduce it. The transition will be gradual, with coexistence of methods for at least 10-15 years.
Could my digital twin data be used against me?
This is one of the biggest risks. Systems use homomorphic encryption so data never leaves the patient's control. GDPR and Brazil's LGPD offer protections, but technology advances faster than regulation.
Will this make medications cheaper?
In theory, significantly. If development costs drop 30-40%, part of that savings should translate to lower consumer prices. However, the pharmaceutical market is notoriously resistant to price reductions.
Conclusion
Digital organ twins are not a distant science fiction promise — they're cutting-edge engineering being built now. If they work as planned, they'll represent nothing less than a rewriting of medicine's rules: from "one-size-fits-all" to "individually tailored," from "test on humans" to "simulate digitally," and from "treat the disease" to "predict and prevent before it appears."
The next revolution in medicine may not happen in a hospital — it may happen inside a server. And the liver that saves your life may be one that doesn't physically exist.





